Thousands of cancer patients in England to benefit from new immunotherapy jab
A new injectable form of pembrolizumab, an immunotherapy treatment for cancer, is set to benefit thousands of patients in England. This treatment can be administered in under two minutes, significantly reducing the time spent in hospitals. The NHS anticipates that this innovation will save over 100,000 hours of preparation and treatment time annually, allowing for more efficient patient care.
At a glance
Location and topic
Region
England
Tag cluster
Trend count
+6 related briefs
What happened
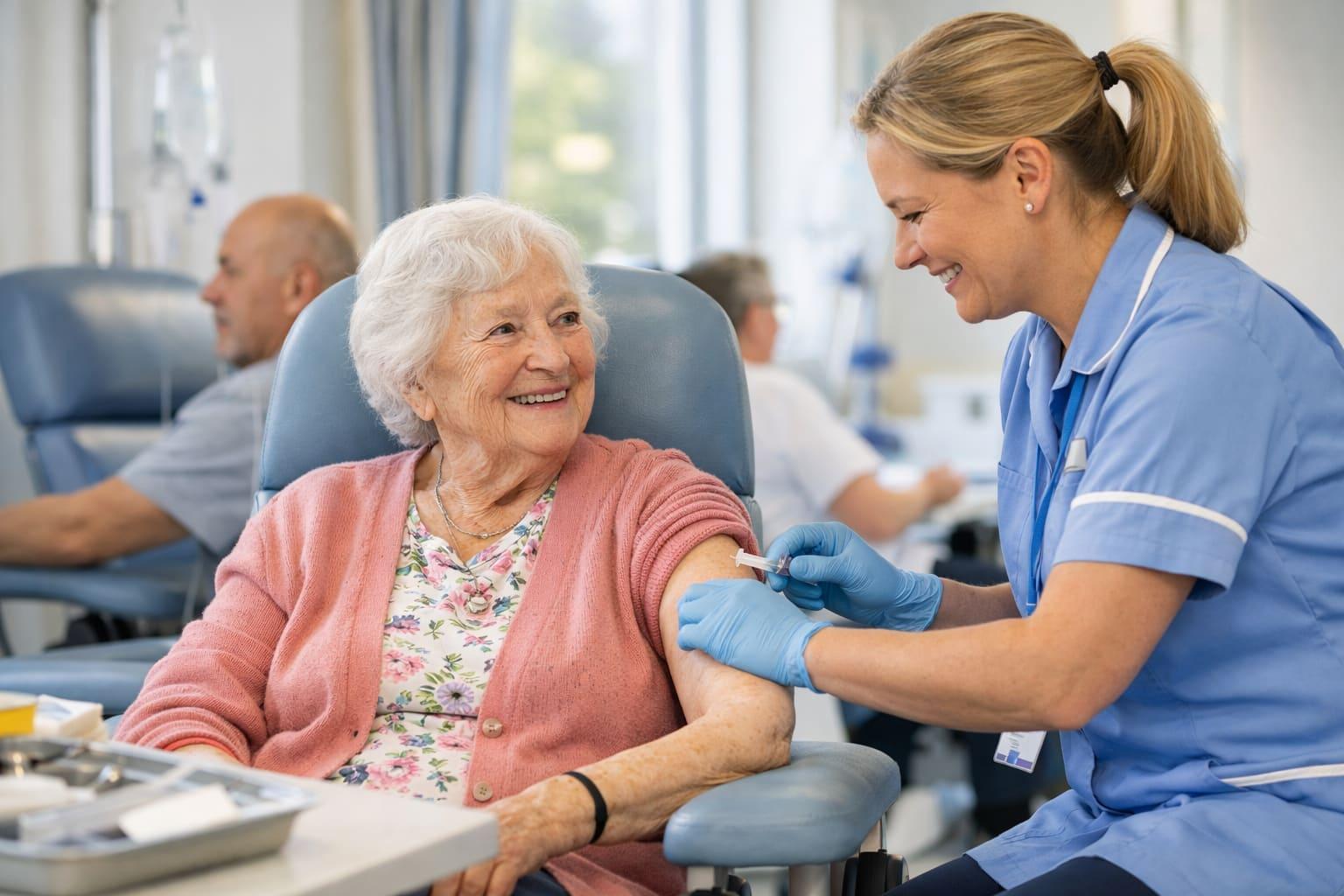
The National Health Service (NHS) in England has announced a significant advancement in cancer treatment with the introduction of a new injectable form of pembrolizumab, an immunotherapy drug that can be administered in under two minutes. This innovative treatment is designed to target several types of cancer by blocking the PD-1 protein, which inhibits the immune system's ability to recognize and attack cancer cells. The previous method of administering pembrolizumab involved an intravenous drip, which required extensive preparation and could take up to two hours per session, making it a time-consuming process for both patients and healthcare providers. With the new injectable version, patients will receive the treatment every three weeks as a one-minute injection or every six weeks as a two-minute injection, significantly reducing the time spent in hospital settings. It is estimated that around 14,000 patients currently receiving the intravenous form of pembrolizumab will transition to this new injectable version, which is expected to save the NHS over 100,000 hours of preparation and treatment time annually. This change not only enhances patient convenience but also allows NHS staff to allocate more time to other patients, thereby improving overall healthcare efficiency. Prof. Peter Johnson, the NHS national clinical director for cancer, emphasized the importance of this treatment in improving patients' quality of life by reducing the time spent in hospital settings. The first patients to receive this new treatment, including 89-year-old Shirley Xerxes from St Albans, have already experienced the benefits of this rapid treatment, highlighting its positive impact on their daily lives. Xerxes expressed her relief at the reduced time spent in the hospital, stating, "I can’t believe how little time it took. It’s made such a difference and gives me more time to live my life, including spending more time gardening."
Why this matters
The introduction of the injectable pembrolizumab represents a significant advancement in cancer care, providing patients with a more efficient and less time-consuming treatment option. This innovation reflects the NHS's commitment to modernizing healthcare and improving patient outcomes. By reducing the burden on healthcare facilities, this new treatment can lead to better overall care for cancer patients, allowing for more timely interventions and potentially better health outcomes. The ability to administer treatment quickly and efficiently not only enhances the patient experience but also alleviates some of the pressures faced by healthcare providers, enabling them to focus on delivering high-quality care to a larger number of patients. This shift towards more patient-centered care is crucial in a healthcare landscape that is increasingly challenged by rising patient numbers and limited resources.
What changed
The rollout of the injectable pembrolizumab marks a significant shift in how cancer treatment is administered in England. Patients will now experience a quicker and more convenient method of receiving their therapy, which is expected to enhance their overall treatment experience. This change is particularly important for those who may have previously found the lengthy intravenous sessions exhausting and time-consuming. The new treatment protocol not only benefits patients by reducing their time in hospital but also allows healthcare providers to optimize their resources and improve service delivery. With the NHS anticipating a substantial reduction in preparation and treatment time, this innovation is expected to free up valuable clinical hours, enabling staff to care for more patients and reduce waiting times for cancer treatments. This transition to a more efficient treatment model is a crucial step in the ongoing effort to modernize cancer care in the UK.
Bigger picture
The introduction of the new injectable pembrolizumab is part of a broader trend in healthcare towards more efficient and patient-centered treatment options. As healthcare systems worldwide face increasing demands, innovations like this are crucial for improving patient care and optimizing resources. The NHS's commitment to modernizing cancer treatment reflects a growing recognition of the need for rapid, effective therapies that can adapt to the needs of patients and healthcare providers alike. This development could serve as a model for other countries looking to enhance their cancer care strategies. The shift towards injectable immunotherapies is indicative of a larger movement within oncology to streamline treatment processes, reduce hospital visits, and improve the overall patient experience. As healthcare systems continue to evolve, the focus on patient convenience and efficiency will likely drive further innovations in cancer treatment and beyond. The success of this new treatment could pave the way for additional advancements in immunotherapy and other areas of cancer care, ultimately leading to better outcomes for patients globally.
History
The development of immunotherapy treatments has revolutionized cancer care over the past few decades. Pembrolizumab, initially introduced as an intravenous treatment, has been a significant advancement in harnessing the immune system to fight cancer. The shift to an injectable form represents a culmination of ongoing efforts to make cancer treatment more accessible and less burdensome for patients. Historically, cancer treatments have often involved lengthy hospital stays and complex administration processes, which can be physically and emotionally taxing for patients. The evolution of immunotherapy, particularly the introduction of drugs like pembrolizumab, has marked a turning point in cancer treatment, offering new hope to patients with various types of cancer. The transition to an injectable format aligns with a broader trend in healthcare towards minimizing patient burden and maximizing treatment efficacy, reflecting a commitment to patient-centered care that has become increasingly important in modern medicine.
Looking Towards the Future
As the new injectable pembrolizumab is rolled out, it will be important to monitor its impact on patient outcomes and NHS efficiency. Observing how this treatment affects waiting times and overall patient satisfaction will provide insights into the future of cancer care in England. Additionally, the response from healthcare providers and patients will be crucial in assessing the long-term viability of this treatment option. Stakeholders will be keen to evaluate whether the anticipated time savings translate into improved access to care for more patients and whether the new treatment leads to better health outcomes. Furthermore, the NHS's ability to manage the transition from intravenous to injectable therapies will be a key factor in determining the success of this initiative. As more patients begin to receive this treatment, tracking its effectiveness and any potential side effects will be essential for ensuring that it meets the needs of those it aims to serve.
Story timeline
Introduction of pembrolizumab
Pembrolizumab was first approved for use in cancer treatment.
Launch of injectable version
The NHS announced the availability of the new injectable form of pembrolizumab.
First patient treatments
Patients began receiving the new injectable treatment on the same day of the announcement.
Sources behind this brief
2 total
The Guardian
Original article detailing the new immunotherapy jab.
NHS England
Official information on NHS cancer treatment innovations.
Further reading on this topic
3 links
NHS England
Official announcement detailing the rollout of the one-minute immunotherapy jab for cancer patients in England.
NHS England
Information on the previous rollout of a 5-minute immunotherapy jab for various cancers.
NHS England
Details about the introduction of a life-extending treatment for advanced cervical cancer in the NHS.
Context zone
England
Context zone
England
On this map
Thousands of cancer patients in England to benefit from new immunotherapy jab
England
No other published articles from this region yet. This marker anchors the story for future regional clustering.